Water's Triple Trick: Solid, Liquid, and Gas All at Once
A short lesson on water property fundamentals.
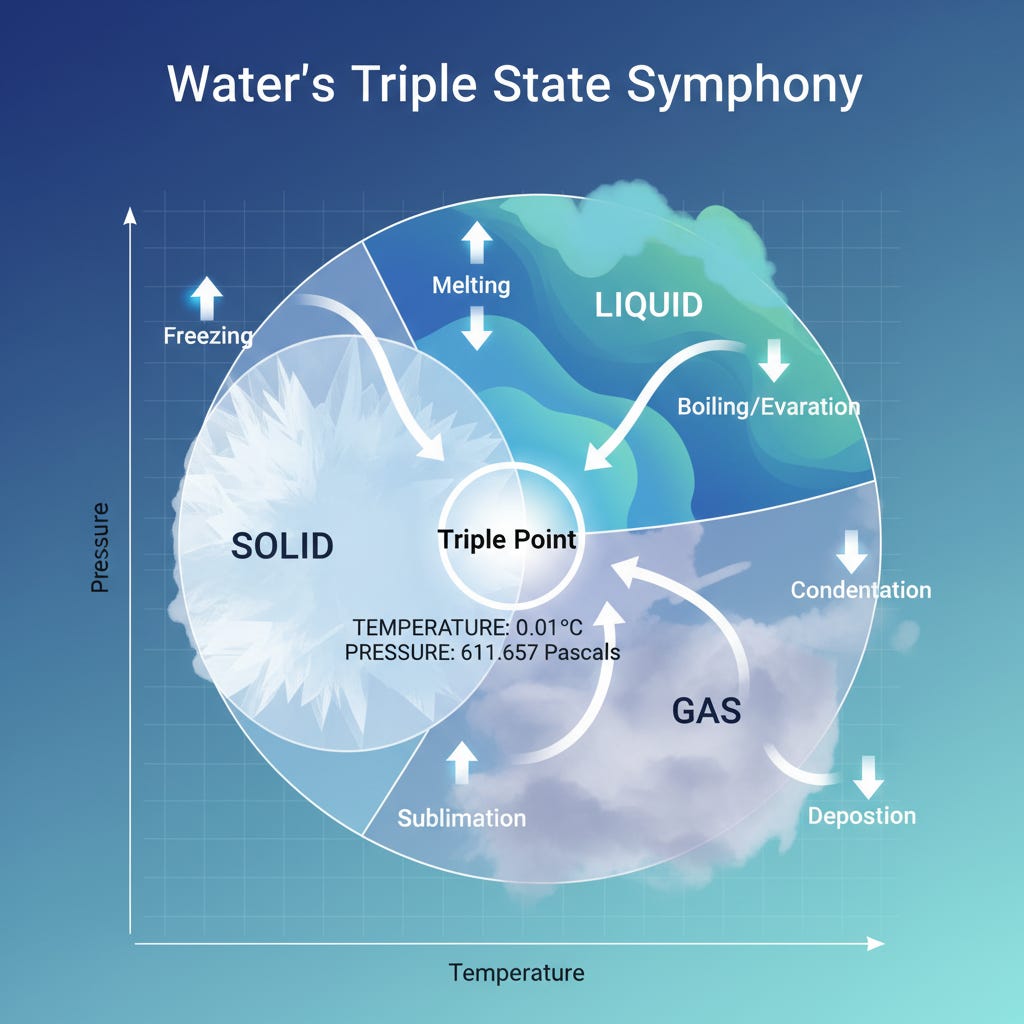
Did you know that water can exist as ice, liquid, and vapor all at the same time? This mind-boggling state occurs at the triple point of water, where temperature and pressure align perfectly to allow water to dance between its three states.
What is the Triple Point of Water?
The triple point of water is a unique condition where water exists simultaneously as a solid, liquid, and gas. It happens at a precise temperature of 0.01°C and a pressure of 611.657 pascals. Imagine a tiny seesaw where each molecule can tip towards being ice, liquid, or vapor!
Historically, the triple point has played a vital role in thermodynamics—the science of heat and energy flow. It helped refine the Kelvin scale, anchoring it to a natural constant [1].
Simply put, under these conditions, water seamlessly transitions between ice, liquid, and vapor. It’s like a molecular ballet where all states are in perfect harmony.
The Science Behind the Triple Point
To grasp the triple point, we turn to phase diagrams, which chart the state of a substance at different temperatures and pressures. These diagrams are like treasure maps for chemists, guiding them through the landscape of phase changes.
Thermodynamic principles, such as equilibrium, are key here. At the triple point, water’s three phases coexist in a delicate balance, achieving a perfect equilibrium [2].
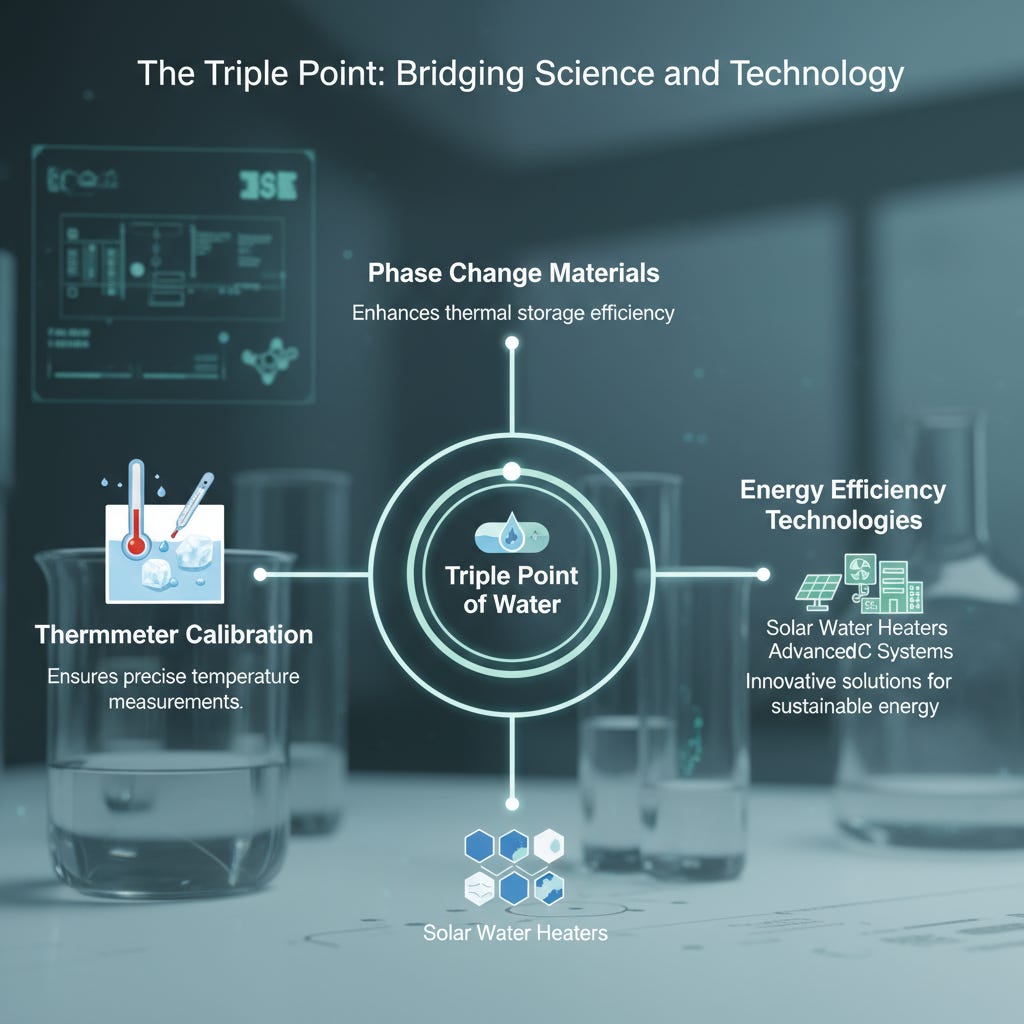
The triple point isn’t just theoretical; it’s a practical tool in science for calibrating thermometers and studying phase transitions.
What Does This Mean for Us?
Beyond the lab, the triple point of water is crucial for scientific precision. It ensures accurate temperature measurements, helping scientists develop technologies like phase change materials for solar water heating systems, which rely on precise thermal properties to boost efficiency [3].
Other substances have fascinating triple points too. For example, carbon dioxide transitions directly from solid to gas under normal conditions, skipping the liquid phase entirely.
This understanding lets us harness natural properties for innovative solutions, from energy efficiency to environmental management.
For example, the triple point sits at the heart of freeze-drying, a technology that keeps vaccines stable without refrigeration and preserves everything from astronaut food to life-saving medicines. The process works by freezing a product solid, then dropping the surrounding pressure below water’s triple point — below that magic 611 pascals — so ice converts directly to vapor without ever becoming a puddle. It’s the molecular equivalent of a disappearing act! This matters enormously for global health: freeze-dried vaccines can be shipped to remote regions without cold chains, dramatically reducing energy use and expanding access to communities that would otherwise go unserved.
On the environmental side, precise knowledge of the triple point turns out to be surprisingly critical in climate monitoring. The sensitive instruments aboard weather balloons and polar research stations must measure temperatures accurately across extreme ranges, and they’re calibrated against triple-point cells — those sealed vessels of ultra-pure water held at perfect equilibrium. Small temperature measurement errors ripple outward into flawed climate models, skewed sea-ice projections, and poorly designed renewable energy systems. In fact, engineers designing more efficient liquefied natural gas (LNG) facilities — which operate at cryogenic temperatures to cut storage volume and carbon footprint — depend on this same triple-point-anchored calibration to minimize energy waste. Nature gave us one precise fixed point, and we’ve built a surprising amount of our clean energy future around it.
Key Takeaways:
- The triple point of water is where it exists as solid, liquid, and gas simultaneously.
- It’s a cornerstone in thermodynamics and vital for precise scientific measurements.
- Understanding it helps develop efficient technologies and offers insights into other substances.
Understanding water’s triple point reveals nature’s intricate balance and the wonders it holds. Who knew a simple molecule like water could teach us so much about the universe’s complexities?
What other everyday substances do you think have surprising behaviors under unique conditions? Share your thoughts or any questions you might have!
References
1. Unknown Author (2006). “Triple point”. Van Nostrand’s Scientific Encyclopedia. DOI: [https://doi.org/10.1002/0471743984.vse8473]
2. Lee, S. (2007). “The Science Behind Triple Point”. Hawley’s Condensed Chemical Dictionary. DOI: [https://doi.org/10.1002/9780470114735.hawley16583]
3. Harris et al. (2023). “Recent developments in phase change material-based solar water heating systems”. International Journal of Thermofluids, Volume 20. DOI: [https://doi.org/10.1016/j.ijft.2023.100359]
Water Weekly is expanding into education. If you’re a teacher or know one, check out my store on Teachers pay Teachers: Water Weekly Education. I have both free and paid resources that cover all sorts of topics in water. Feel free to share with teachers you know, and as always, let me know if you have any feedback.